Request a Tool
Request a Tool
Bond Order Calculator
The bond's order determines the stability of a molecule or ion
Input
Output
Bond Order
0
Formula
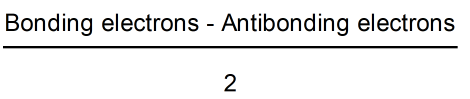
Defination / Uses
The number of electron pairs that form a bond between two atoms is known as bond order. It's used as an indicator of the stability of a bond.
The bond order of a single covalent bond between two atoms is one, a double bond is two, a triple bond is three, and so on. The bond order of two covalently bonded atoms has to be identified.
How to Calculate Bond Order ?
- Enter Number of bonding electrons
- Enter Number of antibonding electrons
- By Subtracting bonding electrons into antibonding electrons and divided by 2
- In Result Display the Bond Order
Use the upper given formula for manual calculations. No sign-up, registration OR captcha is required to use this tool.