Request a Tool
Charles Law Calculator
To calculate Initial Volume, final colume and Initial temperture and final temperture quantities in the Charles law of ideal gas
Input
Output
Input
Output
Input
Output
Input
Output
Formula
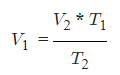
- V1 = Initial Volume
- T1 = Initial Temperature
- V2 = Final Volume
- T2 = Final Temperature
Defination / Uses
Charles's Law is an ideal gas law where at constant pressure the volume of an ideal gas is directly proportional to its absolute temperature.
Uses
Real-life examples of the law used is Hot air balloon, Human lungs, Pool floats, Baking, Tyres and Helium balloon
Formula
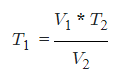
- V1 = Initial Volume
- T1 = Initial Temperature
- V2 = Final Volume
- T2 = Final Temperature
Defination / Uses
Charles's Law is an ideal gas law where at constant pressure the volume of an ideal gas is directly proportional to its absolute temperature.
Uses
Real-life examples of the law used is Hot air balloon, Human lungs, Pool floats, Baking, Tyres and Helium balloon
Formula
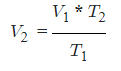
- V1 = Initial Volume
- T1 = Initial Temperature
- V2 = Final Volume
- T2 = Final Temperature
Defination / Uses
Charles's Law is an ideal gas law where at constant pressure the volume of an ideal gas is directly proportional to its absolute temperature.
Uses
Real-life examples of the law used is Hot air balloon, Human lungs, Pool floats, Baking, Tyres and Helium balloon
Formula
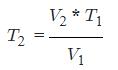
- V1 = Initial Volume
- T1 = Initial Temperature
- V2 = Final Volume
- T2 = Final Temperature
Defination / Uses
Charles's Law is an ideal gas law where at constant pressure the volume of an ideal gas is directly proportional to its absolute temperature.
Uses
Real-life examples of the law used is Hot air balloon, Human lungs, Pool floats, Baking, Tyres and Helium balloon
V1 and V2 be volume at temperature T1 and T2 such that pressure and amount of gas is constant. V1, T1 is volume and temperature at condition 1, and V2, T2 is volume and temperature at condition 2.
Establish the relationship between two equation V1/T1 = k and V2/T2= k. If the volume and temperature for a condition are known, we may use the following method to determine an unknown volume or temperature. Use the upper given formula for manual calculations. No sign-up, registration OR captcha is required to use this tool.