Request a Tool
Request a Tool
Gibbs Energy Calculator
Combines enthalpy and entropy into a single value
Input
Output
Gibbs free energy (ΔG)
0
Formula
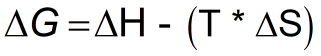
- ΔG = change in Gibbs free energy
- ΔH = change in enthalpy
- ΔS = change in entropy
- T = temperature in Kelvin
Definition / Uses
Gibbs free energy (denoted G) combines enthalpy and entropy into a single value. The change in free energy (ΔG) is equal to the sum of enthalpy plus the product of temperature and entropy of the system. ΔG can predict the direction of the chemical reaction under two conditions: constant temperature and constant pressure. If ΔG is positive, the reaction is nonspontaneous (external energy input required), and if it is negative, the reaction is spontaneous (occurs without external energy input).