Request a Tool
Quick and Easy Oxide Calculator - Free Online Access
An oxide is a binary compound consisting of oxygen and a more electropositive element. Use the Oxide Calculator to determine the oxidation weights of metal oxides.
Input
Output
Oxide Formula
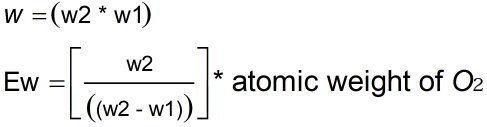
- W2 = Weight of metal oxide
- W1 = Weight of metal
- atomic weight of oxygen = 8
What is an Oxide?
An oxide is a binary compound consisting of oxygen and at least one more electropositive element. These chemical substances are prevalent and easily accessible in nature. Common examples include water (the oxide of hydrogen), silicon dioxide (found in sand and quartz) and carbon dioxide (a byproduct of respiration in animals and plants).
These compounds are formed when elements react with oxygen, often during combustion or oxidation processes.Oxides are found everywhere in nature and play important roles in various chemical reactions.
What is an Oxide Calculator?
An Oxide Calculator is an online tool that helps you find the chemical formula and properties of different oxides. Oxides are chemical compounds that consist of at least one oxygen atom bonded to another element.
Oxide Calculator simplifies complex calculations related to the molecular weight, percentage composition, and stoichiometry of oxide compounds, facilitating more accurate and efficient data analysis.
Why Use Oxide Calculator Tool
Accuracy:
Calculating the properties of oxide compounds manually can be tricky and lead to mistakes, especially with complicated formulas. The Oxide Calculator makes sure you get accurate results every time by doing the calculations automatically.
Efficiency:
This tool makes it faster to figure out different properties of oxides, saving time for researchers, students, and professionals.
User-Friendly:
The calculator is designed to be simple and straightforward, so even those not familiar with chemistry can use it. This user-friendly design means anyone can get the information they need quickly.
Comprehensive Analysis:
Oxide Calculators often include features for detailed analysis, like calculating molecular weight, elemental composition and ratio analysis. These features enable a comprehensive review of the compound's characteristics.
Consistency:
By using a standardized tool, users can ensure that their calculations are consistent, which is crucial for repeatable experiments and comparing data accurately.
Where Oxide Calculator Tool Can Be Used
Material Characterization:
In the characterization of new materials, understanding the oxide composition is fundamental. The Oxide Calculator helps in understanding these materials' properties and potential uses.
Chemical Industry:
The Oxide Calculator is a valuable tool for chemical manufacturers to analyze and regulate the oxide content in different chemical compounds and products. This helps maintain product quality, consistency and compliance with industry standards.
Environmental Analysis:
Environmental scientists utilize these calculators for studying pollutants and natural compounds. They assist in analyzing atmospheric chemistry, soil composition and water quality, aiding in environmental research.
Research and Development:
Researchers and scientists in both academic and industrial research laboratories use the Oxide Calculator for R&D projects. It helps in materials synthesis, characterization, and analysis, supporting the study of material properties, exploration of new applications and advancement of scientific knowledge.
Quality Control:
The Oxide Calculator plays a vital role in manufacturing and production by ensuring quality control. It verifies that product chemical compositions align with specified standards, maintaining product quality and consistency.
How to Use Oxide Calculator
Using an Oxide Calculator typically involves the following steps:
Input Data:
Enter the relevant details of the compound such as the Weight of Metal Oxide (w2) in grams and the Weight of Metal (w1) in grams.
Calculate:
After inputting these values, the tool will automatically calculate the oxidation weight.
View Results:
The tool will show two values like Weight of Oxygen (W) and Equivalent Weight (Ew).
Clear Button:
This button that allows you to clear the input fields and start a new calculation. This button is helpful when you need to perform multiple time calculations or make changes to the input values.
Conclusion:
The Oxide Weight Calculator is an free online tool that shows the oxidation weight of a chemical compound. The weight of oxygen is always half of the metal oxide's weight. Our calculator speeds up the process and gives you results quickly. "Oxide" is a term used for a big group of chemical compounds where oxygen is combined with another element, making it an important part of chemistry.
Metal oxides consist of a metal cation and an oxide anion (O2-). These compounds react with water to create bases and acids that then form salts. You can use the formula provided above for manual calculations and there's no need to sign up, register, or solve captchas to use this tool.