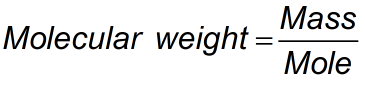Request a Tool
Mole Calculator–Free and Easy-to-Use Online Tool
Mole Calculator helps you to determine the mole, the SI unit used to measure the amount of a substance in chemistry.
Input
Output
Mole Formula
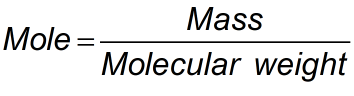
What is a Mole?
In chemistry, a mole is a fundamental unit used to quantify the amount of a substance. It represents 6.022 × 10²³ entities, whether they are atoms, molecules, or ions, and this value is referred to as Avogadro's number. The mole allows scientists to work with macroscopic amounts of substances in a way that relates directly to the atomic or molecular scale.
For example:
- One mole of water (H₂O) contains 6.022×10236.022 \times 10^{23}6.022×1023 molecules of water.
- One mole of carbon atoms weighs 12 grams (its atomic mass in grams)
Moles form the foundation for stoichiometry in chemistry, enabling calculations about the quantities of reactants and products in chemical reactions.
What is a Mole Calculator?
A Mole Calculator is an online tool designed to perform calculations related to moles in chemistry. It enables users to convert between moles and other units such as grams, liters, and molecules, simplifying the problem-solving process. By entering specific variables, the calculator can offer immediate answers, helping both students and professionals save valuable time during experiments or studies.
The tool is particularly essential for stoichiometric calculations, which form the basis for determining the proportions of chemicals needed in reactions. By leveraging mathematical formulas, the calculator provides tremendous assistance to individuals striving to gain a deeper understanding of chemical reactions and molecular relationships.
Why Use a Mole Calculator Tool
Efficiency
In a field where precision is paramount, manually calculating moles can lead to errors. A mole calculator eliminates the risk of miscalculations, enabling users to focus on understanding.
Clarity
The steps involved in mole calculations can often be tedious and confusing, especially for those new to chemistry. A mole calculator presents the necessary calculations in a clear, concise manner, making the learning process smoother.
Accessibility
Chemistry concepts, particularly those involving moles, can seem overwhelmingly complex. A user-friendly mole calculator allows students and professionals alike to access important calculations without needing an extensive math background.
Versatility
Mole calculators often come equipped to handle a variety of calculations related to moles, such as converting moles to grams, grams to moles, moles to volume, and more. This versatility makes it an invaluable asset in numerous chemistry-related tasks.
Where is the Mole Calculator Tool Used
Educational Institutions
They are frequently used in chemistry classes to assist students in learning stoichiometry and chemical calculations.
Research Labs
In academic and industrial research settings, precision in chemical measurements is crucial. Mole calculators streamline the process, enabling researchers to allocate their time to experimental design rather than complex calculations.
Pharmaceutical Industry
Chemists in this field rely on mole calculations for drug formulation, where the precise measurement of reactants can significantly affect the efficacy of a medication.
Environmental Science
Assessing pollutant concentrations and chemical reactions in ecosystems requires accurate mole calculations. Mole calculators assist environmental scientists in their work by providing rapid computation of complex chemical processes.
Food Industry
In food science, understanding chemical reactions during food processing often requires mole calculations to determine ingredient ratios, fermentation processes, and flavor chemistry.
How to Use a Mole Calculator Tool
Using a Mole Calculator is straightforward and involves these simple steps:
Select the Appropriate Calculation
Choose the options from the drop down based on your input. Common functions include converting grams to mole, Molecular Weight and Mass.
Input Data
Enter the known values into the respective fields. For instance, if you’re converting grams to moles, provide the mass of the substance and its molar mass.
Run the Calculation
Once you input the values the tool will process the information and generate an output, which may include the number of moles, grams, or other relevant data.
Interpret Results
Carefully read the results provided by the calculator. Note any additional information such as the molar mass used in the calculation.
Clear Button
This button that allows you to clear the input fields and start a new calculation. This button is helpful when you need to perform multiple time calculations or make changes to the input values.
Conclusion
A Mole Calculator is an indispensable tool in the arsenal of students and professionals navigating the world of chemistry. Its ability to simplify complex calculations promotes efficiency, accuracy, and a clearer understanding of fundamental concepts. From educational contexts to advanced research and industrial applications, the mole calculator proves to be versatile and highly beneficial. By equipping users with instant answers to crucial problems, it fosters a more profound appreciation of the intricate relationships that govern chemical interactions. Armed with this understanding, individuals can confidently approach both theoretical and practical aspects of chemistry, paving the way for innovative discoveries and solutions in various fields.