Request a Tool
Dalton’s Law Partial Pressure Calculator–Simple and Free Online Tool
Use Dalton’s Law Calculator to find total pressure by entering temperature, gas moles, and volume. Simple and accurate!
Input
Output
Daltons Law(Partial pressure) Formula
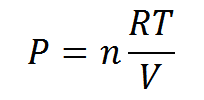
- V = volume
- T = temperature
- Pi = partial pressure of the individual gas
- ni = number of moles of the gas
- R = ideal gas constant with a value of 8.314 J K-1 mol-1
What is Dalton's Law?
Dalton’s Law also known as Dalton’s Law of Partial Pressures stipulates that total pressure exerted by a mixture of gasses that do not chemically interact is the sum of the pressure each gas would exert if it formed the sole constituent. Of particular delicacy is that each gas builds pressure independently of other gasses that may be present, according to the concentration and volume of the respective gas.
What is Dalton's Law(Partial pressure) Calculator?
A Dalton's Law (Partial Pressure) Calculator is an online tool that calculates the total pressure of a gas mixture based on Dalton’s Law of Partial Pressures. This law states that the total pressure exerted by a mixture of gases is equal to the sum of the partial pressures of each individual gas. The calculator uses inputs like temperature, moles of each gas, and volume to compute the overall pressure, making it a valuable resource in chemistry, physics, and engineering. With this tool, users can easily determine how each component gas contributes to the total pressure, simplifying complex gas calculations in labs, classrooms, and research.
This equation is crucial when learning about gasses because it shows how the pressure of the gasses in a mixture are individual of one another.
Why Use a Dalton's Law(Partial pressure) Calculator tool
Accurate Calculations
It offers specific partial pressure data because it can effectively minimize computation inaccuracies
Time-Saving
Fast in calculations particularly required in complex gaseous systems.
User-Friendly
It makes the usage of Dalton’s Law applications, which can be useful for all levels.
Simplifies Complex Calculations
Quickly adds up individual gas pressures to find the total pressure, saving time on manual calculations.
Useful in Labs
Highly important for scientists and laboratory technicians to define the pressures of the gaseous substances.
Convenient
Needs a couple of inputs which gives the output without the need to type away at a keyboard.
Where Can a Dalton's Law(Partial pressure) Calculator tool Be Used
Laboratories
These are used by chemists and researchers to determine partial pressures of gasses that may be in mixture.
Education and Learning
A teaching aid for students learning about gas laws, making it easier to understand and apply Dalton’s Law.
Chemical Engineering
Helps engineers determine pressure levels in reactors, pipelines, and chemical production processes.
Food and Beverage Industry
Useful in processes that involve carbonation or controlled atmospheres, like beverage carbonation or packaging.
Medical Fields
Used in respiratory therapy, anesthesiology, and calculating gas concentrations for safe patient care.
Oil and Gas Industry
To measure gas pressures in natural gas extraction, storage, and transportation, ensuring safe and efficient operations.
How to Use a Dalton's Law(Partial pressure) Calculator tool
Using a Dalton's Law (Partial Pressure) Calculator tool is simple and involves the following steps:
Enter Moles of Gas
Input the number of moles for each gas in the mixture.
Set Temperature
Provide the temperature in Kelvin.
Set Volume
Enter the volume of the container in liters.
Calculate
The tool will automatically calculate the partial pressure of each gas and the total pressure using Dalton’s Law formula.
Review and Adjust
If needed, adjust the input values for temperature, volume, or gas moles to test different scenarios or refine the results.
Clear Button
This button that allows you to clear the input fields and start a new calculation. This button is helpful when you need to perform multiple time calculations or make changes to the input values.
Conclusion
In conclusion, a Dalton's Law (Partial Pressure) Calculator is an essential tool for accurately determining the partial pressures of gasses in a mixture. We're measuring a macroscopic physical property of a large number of gas molecules which aren't visible to the human eye when we perform this.By simplifying complex calculations and ensuring precision, it is invaluable in fields like chemistry, engineering, environmental science, and medicine. This tool aids users in understanding gas behaviors under varying conditions, making it ideal for both educational and professional use. For anyone working with gas mixtures, it provides quick, reliable results, saving time and reducing errors.