Request a Tool
Simplify Acid Weight Calculations - Free Online Tool
Calculate the weight of acid easily with our free online tool. Quick, accurate, and user-friendly, ideal for chemists, students, and professionals needing precise measurements.
Input
Output
Weight of Acid Formula
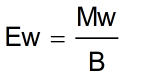
- Mw = Molecular weight of acid
- B = Basicity of acid
- Ew = Equivalent weight of acid
What is a Weight of Acid Calculator?
A Weight of Acid Calculator is an online tool designed to determine the mass or weight of an acid based on its concentration and volume. It simplifies the process by allowing users to input these parameters, typically in units such as moles per liter (Molarity) or grams per liter (Grams per liter), and then calculates the resulting weight of the acid. This tool is essential in chemistry for preparing solutions of precise concentrations, conducting experiments, and ensuring accuracy in chemical reactions and analyses. It is widely used by chemists, educators, students, and professionals working in laboratories and industrial settings.
Weight of Acid Formula
- Mw = Molecular weight of acid
- B = Basicity of acid
- Ew = Equivalent weight of acid
Why to Use a Weight of Acid Calculator?
Accuracy:
Ensures precise measurements of acid mass based on concentration and volume inputs, crucial for preparing solutions with exact chemical compositions.
Efficiency:
Saves time by swiftly calculating acid weights, eliminating manual errors and the need for complex mathematical formulas.
Consistency:
Facilitates consistent preparation of solutions, maintaining experimental reproducibility and reliability in scientific research and industrial processes.
Educational Aid:
Helps students understand the relationship between acid concentration, volume, and mass, enhancing learning in chemistry and laboratory practices.
Practical Applications:
Essential for chemical analysis, environmental monitoring, and quality control in industries such as pharmaceuticals, manufacturing, and agriculture.
Versatility:
Supports calculations for various acids and concentrations, adapting to diverse research and operational requirements.
Accessible:
Available online for free, accessible anytime and anywhere with an internet connection, promoting widespread use and accessibility in educational and professional settings.
Where Does the Weight of Acid Calculator Tool Be Used?
Laboratories:
Used extensively in chemical laboratories for precise preparation of acid solutions used in experiments, titrations, and chemical analyses.
Education:
Employed in educational institutions to teach students about the relationship between acid concentration, volume, and mass, enhancing understanding of chemistry concepts.
Research:
Utilized by researchers in academia and industry for conducting experiments, studying chemical reactions, and developing new formulations.
Environmental Monitoring:
Used in environmental science and monitoring programs to analyze acidity levels in soil, water, and air samples.
Healthcare:
In healthcare facilities for preparing medical solutions and analyzing biological samples in clinical laboratories.
Food and Beverage Industry:
Used for quality assurance and process control in food and beverage production to monitor acidity levels.
DIY and Home Experiments:
Enthusiasts and hobbyists use it for home chemistry experiments and DIY projects involving acids.
How to Use a Weight of Acid Calculator Tool?
Using a Weight of Acid Calculator involves the following steps:
Input Parameters:
Enter the required parameters into the calculator:
- Concentration:Input the concentration of the acid solution. This can be in molarity (M) or grams per liter (g/L).
- Volume:Enter the volume of the acid solution. Ensure the volume is in liters (L) or milliliters (mL).
Selection:
Choose the appropriate units for concentration and volume to match the calculator's requirements.
Calculation:
Click the calculate button or equivalent function on the calculator. The tool will use the relevant formula to compute the weight of the acid.
Result Display:
Instantly view the calculated weight of the acid displayed on the screen. The result is typically shown in grams (g) or kilograms (kg), depending on the calculator.
Verification:
Optionally, verify the computed value by cross-checking with expected values or performing additional calculations if necessary.
Conclusion:
The Weight of Acid Calculator simplifies and enhances accuracy in determining acid mass based on concentration and volume inputs. It streamlines processes in laboratories, supports educational endeavors by illustrating chemical relationships, and aids in research and industrial applications for precise chemical formulations. By providing quick and reliable results, it promotes efficiency and consistency in scientific experiments and quality control measures. Overall, the calculator serves as an indispensable tool in chemistry, facilitating both learning and practical applications across diverse fields.