Request a Tool
Online Boiling Point Elevation Calculator: Simple and Fast Solutions
The boiling point elevation calculator helps you estimate how much the boiling point of a solution increases. It's easy to use and accurate calculations in science.
Input
Output
Boiling Point Elevation Formula

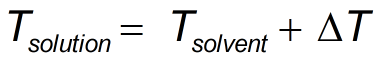
- i = Boiling point of pure solvent (T_solvent)
- Kb = Ebullioscopic constant (K_b)
- m = Molality (m)
What is Boiling Point Elevation?
Boiling Point Elevation means the extent by which the boiling point of a solvent is raised by the addition of a solute to it. This is because the rearrangement of solvated molecules destroys the solvent’s interaction which needs more energy (heat) to boil. It is an essential chemical concept and is used in different fields such as in cooking and manufacturing industries.
What is a Boiling Point Elevation Calculator?
A Boiling Point Elevation Calculator is a tool designed to determine how much the boiling point of a solvent increases when a non-volatile solute is added. This calculator is especially useful in chemistry and various scientific applications, as it simplifies the process of calculating the boiling point elevation based on specific parameters.
This calculator provides quick results without the need for registration, making it ideal for students and professionals.
Why Use the Boiling Point Elevation Calculator tool
Simplifies Calculations
The calculator automates the process of calculating boiling point elevation, saving time and reducing the likelihood of errors that can occur with manual calculations.
Accuracy
The calculator provides precise results based on the input parameters, ensuring reliable outcomes for experiments and practical applications.
Versatile Applications
Useful in various fields such as chemistry, food science, and pharmaceuticals, it helps in formulating solutions and understanding their thermal properties.
Quick Results
Users can quickly obtain boiling point changes, enabling faster decision-making in laboratory and industrial settings.
Parameter Flexibility
The tool allows users to input various parameters (like molality and van't Hoff factor), making it adaptable to different scenarios and solutes.
Where Can the Boiling Point Elevation Calculator tool be used
Chemistry Laboratories
This tool is frequently used in chemistry labs to determine how solutes affect the boiling points of solvents during experiments involving solutions.
Food Science
In food technology, the boiling point elevation calculator is used to adjust cooking times and temperatures for various food products, especially when preparing syrups, sauces, or jams.
Pharmaceutical Industry
Pharmaceutical scientists utilize the calculator to formulate drugs and solutions, ensuring the stability and efficacy of liquid medications under various temperature conditions.
Education
The boiling point elevation calculator serves as an educational tool for students studying colligative properties, allowing them to perform calculations in chemistry coursework and labs.
Textile Industry
The tool can be used in dyeing processes, where the boiling point of water can change due to the presence of dyes and other chemicals, affecting dye uptake in fabrics.
How to Use the Boiling Point Elevation Calculator tool
Enter Values
Input the values for the solute’s molality, the van 't Hoff factor, and the ebullioscopic constant of the solvent.
Calculate
Once all the values input this tool automatically computes the boiling point elevation.
Review Results
View the calculated boiling point elevation and the new boiling point of the solution.
Clear Button
This button that allows you to clear the input fields and start a new calculation. This button is helpful when you need to perform multiple time calculations or make changes to the input values.
Conclusion
Therefore, the Boiling Point Elevation Calculator is a valuable instrument for calculating changes in boiling points of solvents caused by solutes without exerting much time and effort. In the case of the present work, specifically, the choices made to create an appealing and intuitive graphical user interface make the software appropriate for use by both students and professionals in the broad fields of natural sciences to improve understanding of colligative properties. With this calculator in place, users will be able to easily perform a myriad of calculations in everyday learning, engineering, technology and much more.