Request a Tool
Free Molarity Calculator–Fast & Simple Online Tool
Use our Molarity Calculator to easily convert mass concentration to molar concentration, or recalculate grams per ml to moles. Quick, accurate, and free online tool for chemistry calculations.
Input
Output
Formula
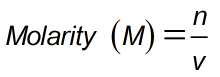
- n = Moles of solute
- v = Liters of solution
What is Molarity?
Molarity is a measure of concentration that indicates the number of moles of a solute dissolved per liter of solution, commonly expressed in units of moles per liter (mol/L). It is a key concept in chemistry, used to quantify the concentration of a substance within a given volume of solvent, making it essential for accurately preparing chemical solutions and conducting reactions. Molarity refers to the amount of solute in solution expressed in a mole of solute per one liter of the solution. Chemistry widely refers to it as the extent of a substance dissolved in a solvent.
What is a Molarity Calculator?
A Molarity Calculator is an online tool designed to help users quickly and accurately determine the molar concentration of a solution. By entering the mass of a solute and the volume of the solution, this calculator converts these values to give the molarity in moles per liter (mol/L). A Molarity Calculator is an interactive utility that enables one to find out the amount of solute in a solution per liter.
This calculator will assist in calculating molarity correctly and will therefore aid in preparation of solutions in laboratories as well as industries.
Why Use a Molarity Calculator Tool
Accurate Calculations
They help in obtaining accurate results especially when it comes to estimating the concentration of solutions without nodal calculations.
Saves Time
Saves time when finding molarity calculations do not require a lot of calculations when preparing particular chemical solutions.
Easy to Use
A number of advantages exist including: simple user interface of the software that can suit students, researchers as well as professionals in chemistry and lab works.
Versatile
Applicable to Another Uses in Chemistries, Biology, and Pharmaceuticals.
Cost-Effective
Minimizes the dependency on dedicated software or time-consuming manual computations, hence it is a useful, cost-friendly instrument.
Where Can a Molarity Calculator Tool Be Used
Educational Institutions
In schools, colleges, and universities in the context of chemical education and as an aid in teaching students factors relating to concentration determination.
Pharmaceuticals
To create precise medication formulation and to test the formulation which will produce an accurate drug solution.
Environmental Testing
In this application the software is used in determining concentrations in water, air, and soil samples as applied in environmental analysis.
Food and Beverage Industry
Scientifically in both; as a component of food science and production for right concentrations of each compound.
Biotechnology
Biotechnological studies and innovation in product development require precise determination of molarity in reactions and processes.
Chemical Engineering
In technical practice to design and improve concentration of solutions in industrial processes.
How to Use a Molarity Calculator Tool
Using a Molarity Calculator tool is simple and involves just a few steps:
Enter the Amount of Solute
Choose between entering the moles or grams of the solute
Enter the Volume of Solution
Specify the volume of the solution in liters or milliliters.
Calculate Molarity
Once you input the values the tool will automatically compute the molarity, giving you the concentration of the solution in moles per liter (mol/L).
Analyze the Results
This button that allows you to clear the input fields and start a new calculation. This button is helpful when you need to perform multiple time calculations or make changes to the input values.
Conclusion
All in all, the Molarity Calculator Tool is quite helpful for anyone who is in need of a quick and efficient way to find out the molarity of a solution. Therefore with the amount of solute and volume of the solution the users are instantly able to get the concentration. This tool makes it easy to prepare various chemical concentrations to be used in laboratories or any other tasks seeing that dilutions are standard.Use the upper given formula for manual calculations. No sign-up, registration OR captcha is required to use this tool.