Request a Tool
Simple and Free Online Concentration Calculator for Accurate Results
Calculate concentration accurately with our free online tool. Easy to use, this calculator helps you determine the amount of solute relative to the total solution. Perfect for students and professionals.
Input
Output
Formula
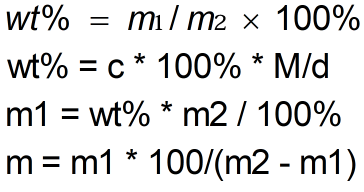
- D = Density
- c = Molarity
- M = Molar Mass
- mass of solution m₂ = 100
- m1 = mass of solute
What is Concentration?
The concentration of a solution refers to the ratio of solute to solvent or the total solution. It describes how much of a substance (solute) is dissolved in a given volume of solvent or solution. Concentration is a key concept in chemistry and can be applied to various types of solutions, not just aqueous ones.
What is a Concentration Calculator?
A Concentration Calculator is an online tool used to determine the concentration of a solute within a solution. It is usually expressed in units such as molarity (moles per liter), weight percent, volume percent, or parts per million (ppm).
The Concentration Calculator helps users perform calculations to find out how concentrated a solution is by providing an accurate and quick method for computing concentration values.
Why Use a Concentration Calculator Tool
Accuracy and Precision
Determining the concentration of a solution can involve complex calculations, especially when dealing with different units and measurements. A Concentration Calculator automates these calculations, reducing the risk of manual errors and ensuring precision in the results.
Efficiency
Manually calculating concentration values can be time-consuming, especially for multiple samples or solutions. The calculator provides instant results, saving time and allowing users to focus on other aspects of their work.
Versatility
The tool can handle various units of concentration, including molarity, weight percent, volume percent and ppm. This versatility makes it useful for a wide range of applications and industries.
Research and Analysis
Researchers use the calculator to ensure that solutions are prepared with the correct concentration, which is crucial for the accuracy of experiments and analyses.
Where Can a Concentration Calculator Tool Be Used
Educational Institutions
The calculator is used in high school and college chemistry courses to help students practice concentration calculations and grasp solution chemistry. It also aids students and educators in preparing solutions with accurate concentrations for laboratory experiments and research projects.
Research Laboratories
Researchers use the calculator to prepare solutions with specific concentrations for experiments, ensuring that the results are reliable and reproducible.In biological research, accurate solution concentrations are necessary for experiments involving enzymes, reagents, and other biological compounds.
Chemical Industry
Manufacturers use the calculator to determine the concentration of active ingredients in chemical products, ensuring consistency and quality in product formulation.The tool is used in quality control processes to verify the concentration of solutions and maintain product standards.
Environmental Science
Environmental scientists use the calculator to determine the concentration of pollutants and contaminants in water samples, aiding in environmental monitoring and regulation . It helps in analyzing soil samples to determine the concentration of nutrients or pollutants, supporting environmental conservation efforts.
How to Use a Concentration Calculator Tool
Using a Concentration Calculator Tool involves a few simple steps:
Enter the Density
Input the density of the solution.
Enter the Molar Mass
Input the molar mass of the solute.
Enter the Molarity
Provide the molarity of the solution.
Perform Calculation
After inputs the numbers this tool automatically performs the concentration calculation.
Review Results
Examine the calculated concentration value provided by the tool.
Clear Button
This button that allows you to clear the input fields and start a new calculation. This button is helpful when you need to perform multiple time calculations or make changes to the input values.
Conclusion:
The Concentration Calculator Tool is a handy resource for accurately determining solute concentrations in solutions. It simplifies complex calculations and improves precision across various fields like education, research, manufacturing, and environmental science.
By automating the process and supporting different units, it helps with preparing solutions, analyzing data, and quality control. Its user-friendly design makes it accessible to students, researchers, and professionals, aiding in accurate and efficient work in many scientific and industrial areas.